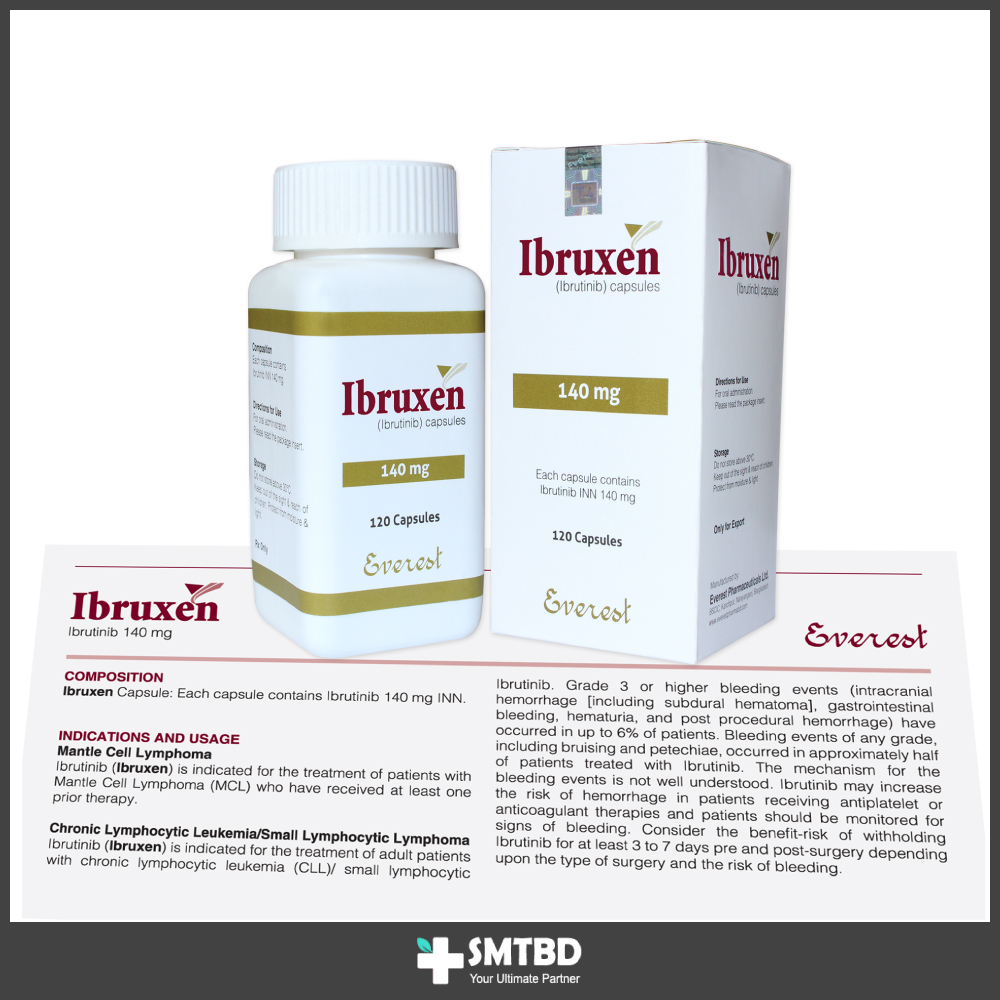
Indications
Mantle Cell Lymphoma: Ibrutinib is indicated for the treatment of patients with Mantle Cell Lymphoma (MCL) who have received at least one prior therapy.
Chronic Lymphocytic Leukemia/Small Lymphocytic Lymphoma: Ibrutinib is indicated for the treatment of patients with chronic lymphocytic leukemia. Chronic Lymphocytic Leukemia/Small Lymphocytic Lymphoma with 17p deletion Ibrutinib is indicated for the treatment of patients with chronic lymphocytic leukemia.
Waldenstrom Macroglobulinemia (WM): Ibrutinib is indicated for the treatment of patients with Waldenstrom Macroglobulinemia (WM).
Marginal Zone Lymphoma: Ibrutinib is indicated for the reatment of patients with marginal zone lymphoma (MZL) who require systemic therapy and have received at least one prior anti-CD 20-based therapy.
Pharmacology
Ibrutinib is a small-molecule inhibitor of BTK. Ibrutinib forms a covalent bond with a cysteine residue in the BTK active site, leading to inhibition of BTK enzymatic activity. BTK is a signaling molecule of the B-cell antigen receptor (BCR) and cytokine receptor pathways. BTK’s role in signaling through the B-cell surface receptors results in activation of pathways necessary for B-cell trafficking, chemotaxis, and adhesion. Nonclinical studies show that ibrutinib inhibits malignant B-cell proliferation and survival in vivo as well as cell migration and substrate adhesion in vitro.
Dosage
Mantle Cell Lymphoma and Marginal Zone Lymphoma: The recommended dose of Ibrutinib for MCL and MZL is 560 mg (four 140 mg capsules) orally once daily until disease progression or unacceptable toxicity.
Chronic Lymphocytic Leukemia/Small Lymphocytic Lymphoma and Waldenstrom Macroglobulinemia (WM): The recommended dose of Ibrutinib for CLL/SLL and WM is 420 mg (three 140 mg capsules) orally once daily until disease progression or unacceptable toxicity. The recommended dose of Ibrutinib for CLL/SLL when used in combination with bendamustine and rituximab (administered every 28 days for up to 6 cycles) is 420 mg (three 140 mg capsules) orally once daily until disease progression or unacceptable toxicity.
Administration
Administer Ibrutinib orally once daily at approximately the same time each day.
Swallow the capsules whole with water. Do not open, break, or chew the capsules.
Interaction
CYP3A Inhibitors: Co-administration with strong and moderate CYP3A inhibitors should be avoided. If a moderate CYP3A inhibitor must be used, Ibrutinib dose should be reduced
CYP3A Inducers: Co-administration with strong CYP3A inducers should be avoided
Contraindications
Hypersensitivity to the active substance or to any of the excipients.
Side Effects
The following adverse reactions are discussed in more detail in other sections of the labeling: Hemorrhage, Infections, Cytopenias, Atrial Fibrillation, Hypertension, Second Primary Malignancies and Tumor Lysis Syndrome. Additional Important Adverse Reactions: Diarrhea, Visual Disturbance.
Pregnancy & Lactation
Pregnancy: Ibrutinib, a kinase inhibitor, can cause fetal harm based on findings from animal studies. In animal reproduction studies, administration of Ibrutinib to pregnant rats and rabbits during the period of organogenesis at exposures up to 2-20 times the clinical doses of 420-560 mg daily produced embryofetal toxicity including malformations. If Ibrutinib is used during pregnancy or if the patient becomes pregnant while taking Ibrutinib, the patient should be apprised of the potential hazard to the fetus. The estimated background risk of major birth defects and miscarriage for the indicated population is unknown.
Lactation: There is no information regarding the presence of Ibruxen or its metabolites in human milk, the effects on the breast fed infant, or the effects on milk production.
Use in Special Populations
Pediatric Use: The safety and effectiveness of Ibruxen in pediatric patients has not been established.
Geriatric Use: Of the 905 patients in clinical studies of Ibruxen, 62% were ≥ 65 years of age, while 21% were ≥75 years of age. No overall differences in effectiveness were observed between younger and older patients. Anemia (all grades) and Grade 3 or higher pneumonia occurred more frequently among older patients treated with Ibrutinib.
Hepatic Impairment: Ibrutinib is metabolized in the liver. In a hepatic impairment study, data showed an increase in Ibruxen exposure. The safety of Ibrutinib has not been evaluated in cancer patients with mild to severe hepatic impairment by Child-Pugh criteria. Monitor patients for signs of Ibrutinib toxicity and follow dose modification guidance as needed. It is not recommended to administer Ibrutinib to patients with moderate or severe hepatic impairment.
Overdose Effects
There is no specific experience in the management of Ibruxen over dose in patients. One healthy subject experienced reversible Grade 4 hepatic enzyme increases (AST and ALT) after a dose of 1680 mg. Closely monitor patients who ingest more than the recommended dosage and provide appropriate supportive treatment.
Therapeutic Class
Targeted Cancer Therapy
Storage Conditions
Store in a dry place below 30°C, protect from light. Keep out of the reach of children.
Manufacturer: Everest Pharmaceuticals Ltd.
For More Product please visit


Reviews
There are no reviews yet.