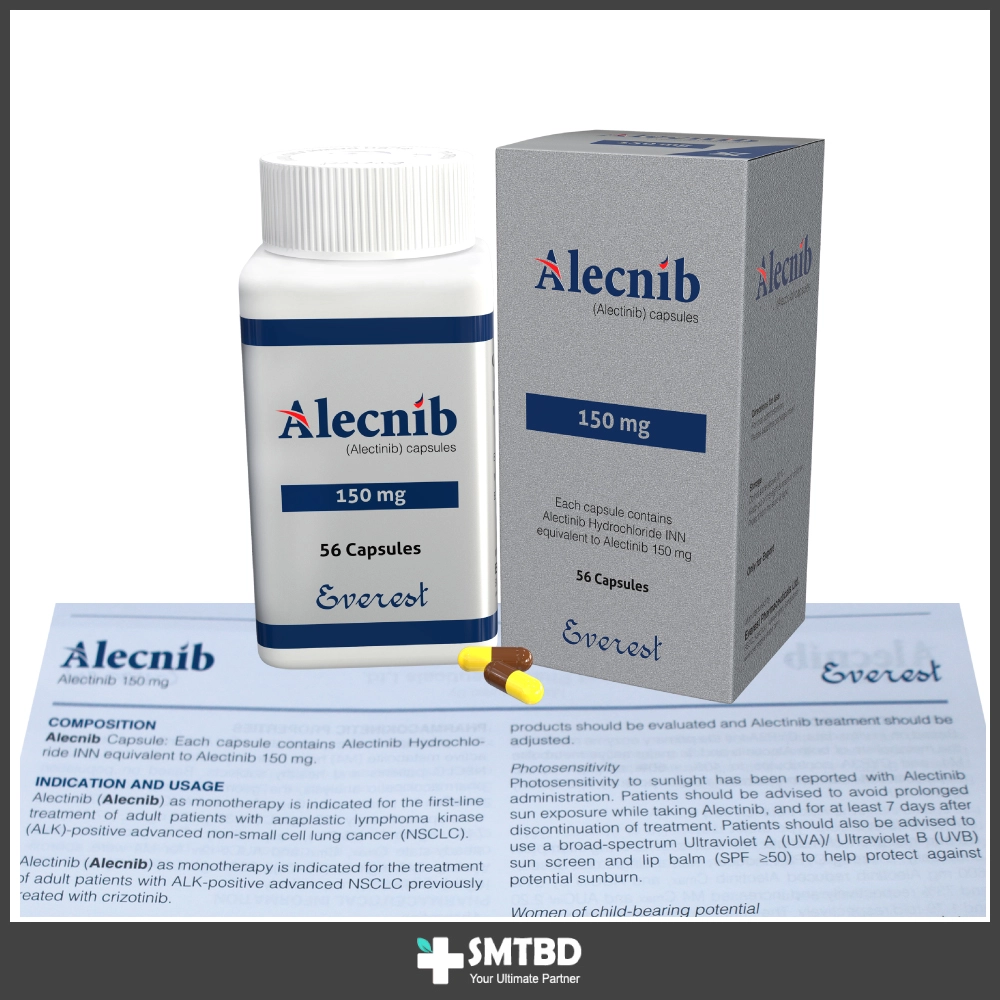
Indications
Alectinib indicated for the treatment of patients with anaplastic lymphoma kinase (ALK)-positive, locally advanced or metastatic non-small cell lung cancer (NSCLC) who have progressed on or are intolerant to crizotinib.
Pharmacology
Alectinib is a highly selective and potent ALK and RET tyrosine kinase inhibitor. In nonclinical studies, inhibition of ALK tyrosine kinase activity led to blockage of downstream signaling pathways including STAT 3 and PI3K/AKT and induced tumor cell death (apoptosis).
Alecnib demonstrated in vitro and in vivo activity against mutant forms of the ALK enzyme, including mutations responsible forresistance to crizotinib. The major metabolite of alectinib (M4) has shown similar in vitro potency and activity.
Based on non clinical data, Alecnib is not a substrate of p-glycoprotein (P-gp) or breast cancer resistance protein (BCRP), which are both efflux transporters in the blood brain barrier, and is therefore able to distribute into and be retained within the central nervous system. Alecnib induced tumor regression in non clinical mousexeno graft models, including antitumor activity in the brain, and prolonged survival in intracranial tumor animal models.
Dosage
The recommended dose of Alectinib is 600 mg (four 150 mg capsules) given orally, twice daily (total daily dose of 1200 mg). Patients with underlying severe hepatic impairment should receive a dose of 450 mg given orally twice daily (total daily dose of 900 mg). Alecnib hard capsules should be taken with food, swallowed whole and must not be opened or dissolved.
Administration
Duration of Treatment: It is recommended that patients are treated with Alectinib until disease progression or unmanageable toxicity.
Dose Modifications: Management of adverse events may require temporary interruption, dose reduction, or discontinuation of treatment with Alecnib. The dose of Alecnib should be reduced in steps of 150 mg twice daily based on tolerability. Alectinib treatment should be permanently discontinued if patients are unable to tolerate the 300 mg twice-daily dose.
Interaction
In vitro studies indicate that neither alectinib nor its major active metabolite (M4) inhibits CYP1A2, CYP2B6, CYP2C9, CYP2C19, or CYP2D6 at clinically relevant concentrations. Alectinib and M4 show weak time-dependent inhibition of CYP3A4. In vitro, Alecnib exhibits a weak induction potential of CYP3A4 and CYP2B6 at clinical concentrations. Results from a clinical drug-drug interaction study in ALK-positive NSCLC patients demonstrated that multiple doses of alectinib had no influence on the exposure of midazolam, a sensitive CYP3A substrate. Therefore, no dose adjustment is required for co-administered CYP3A substrates. Although in vitro studies indicate that alectinib is an inhibitor of CYP2C8, physiologically based pharmacokinetic (PBPK) modeling supports that at clinically relevant concentrations Alecnib does not have the potential to increase plasma concentrations of co-administered substrates of CYP2C8.
Contraindications
Alectinib is contraindicated in patients with a known hypersensitivity to alectinib or any of the excipients.
Side Effects
The most common adverse drug reactions were constipation, edema including peripheral, generalized, eyelid, periorbital; myalgia (31% including myalgia and musculoskeletal pain), nausea, increased bilirubin (21% including increased blood bilirubin, hyperbilirubinemia and increased bilirubin conjugated), anemia (20%, including anemia and hemoglobin decreased), and rash (20%, including rash, rash maculopapular, dermatitis acneiform, erythema, rash generalized, rash papular, rash pruritic and rash macular).
Pregnancy & Lactation
Pregnancy: Women of childbearing potential must be advised to avoid pregnancy while on Alectinib. No clinical studies of Alectinib in pregnant women have been performed. Based on its mechanism of action, Alectinib may cause fetal harm when administered to a pregnant woman. Female patients or women who are partners of male patients receiving Alectinib, who become pregnant while taking Alectinib or during the 3 months following the last dose of Alectinib must contact their doctor and should be advised of the potential harm to the fetus.
Lactation: It is not known whether Alectinib is excreted in human breast milk. No studies have been conducted to assess the impact of Alectinib on milk production or its presence in breast milk. As many drugs are excreted in human milk and because of the potential harm to the infant, mothers should be advised against breastfeeding while receiving Alectinib.
Contraception: Female patients of child-bearing potential, or women of child-bearing potential who are partners of male patients receiving Alectinib, must use highly effective contraceptive methods during treatment and for at least 3 months following the last dose of Alectinib
Precautions & Warnings
Interstitial lung disease (ILD)/pneumonitis: Cases of ILD/pneumonitis have been reported in clinical trials with Alectinib. Patients should be monitored for pulmonary symptoms indicative of pneumonitis. Alectinib should be immediately interrupted in patients diagnosed with ILD/pneumonitis and should be permanently discontinued if no other potential causes of ILD/pneumonitis have been identified
Hepatotoxicity: Elevations in alanine amino transferase (ALT) and aspartate amino transferase (AST) greater than 5 times the upper limit of normal (ULN) as well as bilirubin elevations of more than 3 times the ULN occurred in patients in pivotal clinical trials with Alectinib. The majority of these events occurred during the first 3months of treatment. In the pivotal Alectinib clinical trials it was reported that three patients with Grade 3‒4 AST/ALT elevations had drug-induced liver injury. Concurrent elevations in ALT or AST greater than or equal to three times the ULN and total bilirubin greater than or equal to two times the ULN, with normal alkaline phosphatase, occurred in 1 patient treated in Alectinib clinical trials.
Use in Special Populations
Pediatric use: The safety and efficacy of Alecnib in children and adolescents (<18 years) have not been studied.
Geriatric use: No dose adjustment of Alecnib is required in patients≥65 years of age.
Renal impairment: No dose adjustment is required in patients with mild or moderate renal impairment. alectinib has not been studied in patients with severe renal impairment, however, since Alecnib elimination via the kidney is negligible, no dose adjustment is required inpatients with severe renal impairment
Hepatic impairment: No dose adjustment is required in patients with underlying mild or moderate hepatic impairment. Patients with underlying severe hepatic impairment should receive a dose of 450 mg given orally twice daily (total daily dose of 900 mg)
Therapeutic Class
Anti neoplastic preparations, Protein kinase inhibitor
Storage Conditions
Keep below 30°C temperature, away from light & moisture. Keep out of the reach of children.
Manufactured by : Everest Pharmaceuticals Ltd.
For More Product Please Visit


Reviews
There are no reviews yet.